Gaseous StateHard
Question
X mL of H2 gas effuses through a hole in container in 5 sec. The time taken for the effusion of the same volume of the specified below under identical conditions is :
Options
A.10 sec : He
B.20 sec : O2
C.25 sec : CO
D.55 sec : CO2
More Gaseous State Questions
1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is...At constant pressure which of the following does not represent Charle′s law ?...Oxygen and cyclopropane at partial pressures of 570 torr and 170 torr respectively are mixed in a gas cylinder. What is ...If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with inc...For the non-zero values of force of attraction between gas molecules, gas equation will be :...
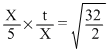 for H2 and O2
for H2 and O2