Gaseous StateHard
Question
Oxygen and cyclopropane at partial pressures of 570 torr and 170 torr respectively are mixed in a gas cylinder. What is the ratio of the number of moles of cyclopropane to the number of moles of oxygen?
Options
A.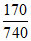 = 0.23
= 0.23
B.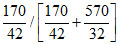 = 0.19
= 0.19
C.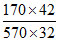 = 0.39
= 0.39
D.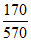 = 0.30
= 0.30
More Gaseous State Questions
Calculate the radius of He atoms if its Vander Waal′s constant ′b′ is 24 ml mol-1....A substance AxBy crystallizes in a face-centred cubic (fcc) Lattice in which atoms ′A′ occupy each corner of...Compressibility factor for H2 behaving as real gas is :...A vessel of volume 5 litre contains 1.4 g of nitrogen at a temperature 1800 K. The pressure of the gas if 30% of its mol...Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted b...