SolutionHard
Question
A solution containing 28 g of phosphorus in 315 g CS2 (b.p. 46.3oC) boils at 47.98oC. If Kb for CS2 is 2.34 K kg mol-1. The formula of phosphorus is (at. mass of P = 31).
Options
A.P6
B.P4
C.P3
D.P2
Solution
ᐃTb = m Kb = 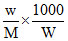 × Kb
× Kb
ᐃTb = 47.98 - 46.3 = 1.68
1.68 =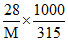 × 2.28
× 2.28
M =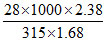 = 125.92
= 125.92
Atomicity =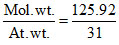 = 4.02
= 4.02
So. Molecule is = P4 .
ᐃTb = 47.98 - 46.3 = 1.68
1.68 =
M =
Atomicity =
So. Molecule is = P4 .
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of benzene at 30oC is 121.8 mm. By adding 15g of non-volatile solute in 250 g of benzene, its molecu...Elevation in b.p. of an aqueous urea solution is 0.52o. (Kb = 0.52o mol-1 kg) Hence, mole-fraction of urea in this solut...The process of getting fresh water from sea water is known as...100 ml aq. solution of glucose with osmotic pressure 1.2 atm at 25oC is mixed with 300 ml aq. solution of urea of 2.4 at...For 1 molal aqueous solution of the following compounds, which one will show the highest freezing point ?...