SolutionHard
Question
The vapour pressure of benzene at 30oC is 121.8 mm. By adding 15g of non-volatile solute in 250 g of benzene, its molecular weight of solute is :
Options
A.156.6g
B.267.4 g
C.356.3 g
D.467.4 g
Solution
According to Raoult′s law,
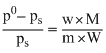
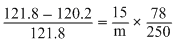
∴ m =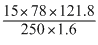 = 356.265 g
= 356.265 g
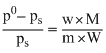
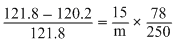
∴ m =
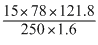 = 356.265 g
= 356.265 gCreate a free account to view solution
View Solution FreeMore Solution Questions
The van’t Hoff factor for Ba(NO3)2 solution is 2.74. The degree of dissociation of salt is...Vapour pressure of a solution of 5 g of nonelectrolyte in 100 g of water at a particular temperature is 2985 N/m2. If th...The product of hydrolysis of (P) and (Q) can be distinguished by -...0.450 g of urea (mol.wt. 60) in 22.5 g of water show 0.170oC of elevation in boiling point. The molal elevation constant...Which of the following is a correct statement -...