SolutionHard
Question
Elevation in b.p. of an aqueous urea solution is 0.52o. (Kb = 0.52o mol-1 kg) Hence, mole-fraction of urea in this solution is :
Options
A.0.982
B.0.567
C.0.943
D.0.018
Solution
As ᐃTb = molality × Kb
0.52 = m × 0.52
molality = 1 mol kg-1
∴ urea = 1 mol
moles of water =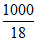 = 55.55
= 55.55
mole fraction of urea =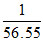 = 0.018
= 0.018
0.52 = m × 0.52
molality = 1 mol kg-1
∴ urea = 1 mol
moles of water =
mole fraction of urea =
Create a free account to view solution
View Solution FreeMore Solution Questions
Water and chlorobenzene are immiscible liquids. Their mixture boils at 90°C under a reduced pressure of 9.031 × 104 Pa. ...Which of the following aqueous solution will have the highest boiling point?...Heptane and octane form ideal solution. At 373 K, the vapour pressures of the pure liquids are 106 kPa and 46 kPa, respe...Which one of the following statements is false?...Which of the following is less than zero for ideal solutions ?...