SolutionHard
Question
Consider equimolal aqueous solutions of NaHSO4 and NaCl with ᐃTb and ᐃT′b as their respective boiling point elevations. The value of  will be :
will be :
Options
A.1
B.1.5
C.3.5
D.2/3
Solution
As m → 0 (infinite dilution) both electrolytes will be completely dissociated so
NaCl ⇋ Na+ + Cl-
NaHSO4 ⇋ Na+ + H+ + SO42-.
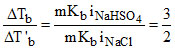 = 1.5
= 1.5
NaCl ⇋ Na+ + Cl-
NaHSO4 ⇋ Na+ + H+ + SO42-.
Create a free account to view solution
View Solution FreeMore Solution Questions
A quantity of 2 g of C6H5COOH dissolved in 25 g of benzene shows a depression in freezing point equal to 1.96 K. Molar d...If 100 mL of 1 M H2SO4 solution is mixed with 100 mL of 98% (W/W) of H2SO4 solution (d = 0.1 g mL), then...A liquid solution is formed by mixing 10 moles of aniline and 20 moles of phenol at a temperature where the vapour press...The vapor pressures of benzene, toluene and a xylene are 75 Torr, 22 Torr and 10 Torr at 20oC. Which of the following is...Benzene and naphthalene forms an ideal solution at room temperature. For this process, the true statement(s) is(are)...