SolutionHard
Question
If 100 mL of 1 M H2SO4 solution is mixed with 100 mL of 98% (W/W) of H2SO4 solution (d = 0.1 g mL), then
Options
A.Concentration of solution becomes half.
B.Volume of solution becomes 200 mL.
C.Mass of H2SO4 in the solution is 98 g.
D.Mass of H2SO4 in the solution is 19.6 g.
Solution
100 mL of 1 M H2SO4 + 100 mL (98%, d = 0.1) H2SO4
⇒ 100 × 1 M + 100 mL ×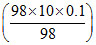
⇒ 100 × 1 M + 100 × 1 M
∴ [H2SO4] =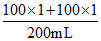 = 1 M = 98 g/1000 mL
= 1 M = 98 g/1000 mL
Mass of H2SO4 =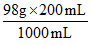 = 19.6 g
= 19.6 g
Concentration of each component becomes half of the initial
⇒ 100 × 1 M + 100 mL ×
⇒ 100 × 1 M + 100 × 1 M
∴ [H2SO4] =
Mass of H2SO4 =
Concentration of each component becomes half of the initial
Create a free account to view solution
View Solution FreeMore Solution Questions
A 0.50 molal solution of ethylene glycol in water is used as coolant in a car. If the freezing point constant of water i...Relative decrease in vapour pressure of an aqueous solution containing 2 moles [Cu(NH3)3Cl] Cl in 3 moles H2O is 0.50. O...A Solution of sucrose (molar mass = 342 gm-mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. ...Freezing point lowering expression is ᐃTf = Kfm (molality)Which of the following assumptions are considered for th...A sample of air is saturated with benzene (vapor pressure = 100 mm Hg at 298 K) at 298K, 750mm Hg pressure. If it is iso...