Gaseous StateHard
Question
For a real gas the P-V curve was experimentally plotted and it had the following appearance. With respect to liquifaction. Choose the correct statement.
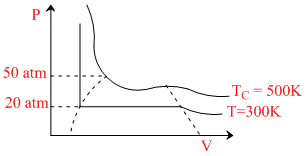

Options
A.at T = 500 K, P = 40 atm, the state will be liquid.
B.at T = 300 K, P = 50 atm, the state will be gas
C.at T < 300 K, P > 20 atm, the state will be gas
D.at 300 K < T < 500 K, P > 50 atm, the state will be liquid.
Solution
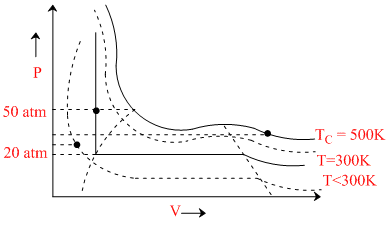
(a) at T = 500 K, P = 40 atm corresponds to ′a′ substance - gas
(b) at T = 300 K, P = 50 atm corresponds to ′b′ substance - liquid
(c) at T < 300 K, P > 20 atm corresponds to ′c′ substance - liquid
(d) at T < 500 K, P > 50 atm corresponds to ′d′ substance - liquid
So, Answer (D)
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Consider the following statements: The coefficient B in the virial equation of state(i) is independent of temperature(ii...Which of the following is not the correct set of pressure and volume at constant temperature and constant moles of gas ?...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted b...The crystal system of a compound with unit cell dimensions ″a = 0.387, b = 0.387 c = 0.504nm and α = β =...