SolutionHard
Question
Osmotic pressure of 30% solution of glucose is 1.20 atm and that of 3.42% solution of cane sugar is 2.5 atm. The osmotic pressure of the mixture containing equal volumes of the two solutions will be
Options
A.2.5 atm
B.3.7 atm
C.1.85 atm
D.1.3 atm.
Solution
πfVf = π1V1 + π2V2
πf =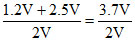
πf = 1.85 atm.
πf =
πf = 1.85 atm.
Create a free account to view solution
View Solution FreeMore Solution Questions
Freezing point of an aqueous solution is (-0.186)oC. Elevation of boiling point of the same solution is Kb = 0.512 0C, K...Addition of 4.0 g of a non-volatile solute to 54.0 g water results in a lowering of vapour pressure by 1.0%. What is the...There are some of the characteristics of the supersaturated solutionI : Equilibrium exists between solutions and solid s...Liquids A and B form an ideal solution. The plot of $\frac{1}{X_{A}}$ (Y-axis) versus$\frac{1}{Y_{A}}$ (X-axis) (where X...A complex of iron and cyanide ions is 100% ionised at 1m (molal). If its elevation in b.p. is 2.08. Then the complex is ...