SolutionHard
Question
A complex of iron and cyanide ions is 100% ionised at 1m (molal). If its elevation in b.p. is 2.08. Then the complex is (Kb = 0.52o mol-1 kg) :
Options
A.K3[Fe(CN)6]
B.Fe(CN)2
C.K4[Fe(CN)6]
D.Fe(CN)4
Solution
ᐃTb = ikb m
so i =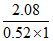 = 4
= 4
so the compex is K3 [Fe(CN6)]
K3 [Fe(CN)6] ⇋ 3 K+ + [Fe(CN)6]3-
so i =
so the compex is K3 [Fe(CN6)]
K3 [Fe(CN)6] ⇋ 3 K+ + [Fe(CN)6]3-
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following 0.1 M aqueous solution will have the lowest freezing point ?...The normal freezing point of nitrobenzene is 278.82 K. A non-volatile solute is dissolved in it and a solution of molali...A solution of sulphuric acid in water exhibits :...Which loses weight on exposure to the atmosphere:-...What is pH of the solution when 100 ml of 0.1M HCl is mixed with 100 ml of 0.1M CH3COOH....