SolutionHard
Question
A solution of a substance containing 1.05 g per 100 mL. was found to be isotonic with 3% glucose solution. The molecular mass of the substance is :
Options
A.31.5
B.6.3
C.630
D.63
Solution
For isotonic solution π1 = π2 ; C1 = C2 ; n1 = n2
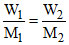 ⇒
⇒ 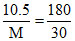 ⇒ M =
⇒ M = 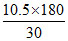 = 63 Ans.
= 63 Ans.
Create a free account to view solution
View Solution FreeMore Solution Questions
A complex containing K+, Pt(IV) and Cl- is 100% ionised giving i = 3. Thus, complex is :...We have 100 mL of 0.1 M KCl solution. To make it 0.2 M,...Electrolysis of dilute aq. NaCl solution was carried out by pasing 10 mA current. The time required to liberate 0.01 mol...Pressure cooker reduces cooking time because...The vapour pressure of water at 20oC is 17.54 mmHg. What will be the vapour pressure of the water in the apparatus shown...