SolutionHard
Question
The vapour pressure of water at 20oC is 17.54 mmHg. What will be the vapour pressure of the water in the apparatus shown after the piston is lowered, decreasing the volume of the gas above the liquid to one half of its initial volume (assume temperature constant).
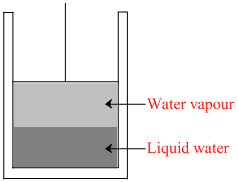

Options
A.8.77 mmHg
B.17.54 mmHg
C.35.08 mmHg
D.between 8.77 and 17.54 mmHg
Solution
Vapour pr. depends on temperature not volume.
Create a free account to view solution
View Solution FreeMore Solution Questions
Of the following 0.10 m aqueous solutions, which one will exhibit the largest freezing point depression?...A 0.2 molal aqueous solution of a weak acid (HX) is 20 per cent ionised. The freezing point of this solution is (Given k...Consider following terms (m = molality) :I : mKb ; II : mKbiIII : IV : Kb Terms which can be expressed in degree (temper...Mixture of volatile components A and B has total vapour pressure (in Torr) p = 254 - 119 xA where xA is mole fraction of...Consider a binary mixture of volatile liquids. If at XA = 0.4 the vapour pressure of solution is 580 torr then the mixtu...