SolutionHard
Question
We have 100 mL of 0.1 M KCl solution. To make it 0.2 M,
Options
A.evaporate 50 mL water
B.evaporate 50 mL solution
C.add 0.1 mol KCl
D.add 0.01 mol KCl
Solution
nitial moles of KCl = 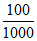 × 0.1 = 0.01
× 0.1 = 0.01
Let x moles of KCl are added , so
0.2 =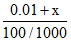 or x = 0.01
or x = 0.01
Let x moles of KCl are added , so
0.2 =
Create a free account to view solution
View Solution FreeMore Solution Questions
Select incorrect statement :...A sample of air is saturated with benzene (vapor pressure = 100 mm Hg at 298 K) at 298K, 750mm Hg pressure. If it is iso...Which of the following liquid pairs shows a positive deviation from Raoult′s law?...PtCl4. 6H2O can exist as a hydrated complex 1 molal aq. solution has depression in freezing point of 3.72o. Assume 100% ...Barium ions, CN- and Co2+ form an ionic complex. If that is supposed to be 75% ionised in water with vant Hoff factor (i...