Coordination CompoundHard
Question
Both geometrical and optical isomerism are shown by :
Options
A.[Co(en)2Cl2]+
B.[Co(NH3)5Cl]2+
C.[Co(NH3)4Cl2]+
D.[Cr(OX)3]3-
Solution
[Co(en)2Cl2]+ show geometrical isomerism and its only cis-form show optical isomerism.
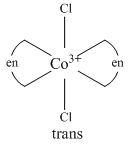
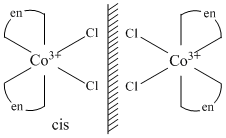
(B) [Co(NH3)5Cl]2+ exists only in one form.
(C)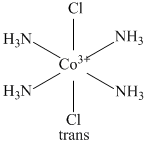
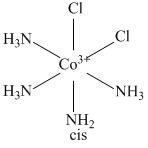
It does not shows optical isomerism because of the presence of plane of symmetry and centre of symmetry.
(D) [Cr(OX)3]3- shows optical isomerism but does not show geometrical isomerism because of the same type of ligands.


(B) [Co(NH3)5Cl]2+ exists only in one form.
(C)


It does not shows optical isomerism because of the presence of plane of symmetry and centre of symmetry.
(D) [Cr(OX)3]3- shows optical isomerism but does not show geometrical isomerism because of the same type of ligands.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Among TiF62–, CoF63– , Cu2Cl2 and – NiCl42– the colourless species are:...F2 is formed by reaccting by reaching K2MnF6 with :...(CFSE = Crystal Field stasilisation energy )The correct relation of x1 and x2 are :-...Of the following configurations, the optical isomers are :...50 ml of 0.2 M solution of a compound with empirical formula CoCl3.4NH3 on treatment with excess of AgNO3(aq) yields 1.4...