Coordination CompoundHard
Question
F2 is formed by reaccting by reaching K2MnF6 with :
Options
A.SbF5
B.MnF3
C.KSbF6
D.MnF4
Solution
SbF5, being the stronger lewis acid, displaces the weaker one (MnF4) from its salt. MnF4, being unstable, decomposes to give 4 MnF4 and fluorine.
K2MnF6 + 2SbF5 → 2SbF6 + MnF4
Unstable
MnF4 → MnF3 +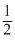 F2
F2
K2MnF6 + 2SbF5 → 2SbF6 + MnF4
Unstable
MnF4 → MnF3 +
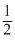 F2
F2Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
To obtain silver from silver amalgam, it heated in vessel which is made of -...Which of the following pairs of compounds are positional isomers ?...The geometry of [NiCI4]2- and [NiCI2 (PMe3)2] are :...On treatment of [Ni(NH3)4]2+ with concentrated HCl, two compounds I and II having the same formula, [NiCl2(NH3)2] are ob...Which of the following is correct about? Tetraamminedithiocyanato-scobalt(III) tris(oxalato)cobaltate(III)...