Coordination CompoundHard
Question
50 ml of 0.2 M solution of a compound with empirical formula CoCl3.4NH3 on treatment with excess of AgNO3(aq) yields 1.435 g of AgCl. Ammonia is not removed by treatment with concentrated H2SO4. The formula of the compound is:
Options
A.Co(NH3)4Cl3
B.[Co(NH3)4Cl2]Cl
C.[Co(NH3)4Cl3
D.[CoCl3(NH3)]NH3
Solution
mole of complex = 50 × 0.2 = 0.01 and mole of AgCl = 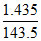 = 0.01
= 0.01
n Ag+ = n Cl-
∴ 1 mole complex = 1 mole AgCl
n Ag+ = n Cl-
∴ 1 mole complex = 1 mole AgCl
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The complex ion in the compound Cs[MnF4(H2O)2] is octahedral and high spin. The name of the compound and the d-orbital s...Which of the following can exhibit geometrical isomerism?...Which one the following has largest number of isomers?...Which one of the following has an optical isomer ? (en = ethylenediamine)...Which of the following statement(s) is /are correct ?...