Coordination CompoundHard
Question
The geometry of [NiCI4]2- and [NiCI2 (PMe3)2] are :
Options
A.both square planar.
B.tetrahedral and square planar respectively.
C.both tetrahedral.
D.square planar and tetrahedral respectively.
Solution
In both complexes the oxidation state of Ni is +2. In [NiCl4]2- the Cl- is a weak field ligand, so it does not compel for pairing. Then,
[NiCI4]2-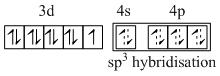
Hence NiCI4 is paramagnetic and tetrahedral.
In [NiCI2 (PMe3)2] the trimethyl phosphine is a strong field ligands; so it compels for the pairing of electrons. Then,
[NiCI2 (PMe3)2]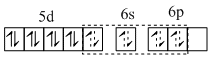
Hence [NiCI2 (PMe3)2] is diamagnetic and square planar.
[NiCI4]2-
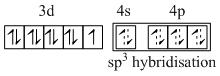
Hence NiCI4 is paramagnetic and tetrahedral.
In [NiCI2 (PMe3)2] the trimethyl phosphine is a strong field ligands; so it compels for the pairing of electrons. Then,
[NiCI2 (PMe3)2]

Hence [NiCI2 (PMe3)2] is diamagnetic and square planar.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following electronic arrangement gives the highest value of the magnetic moment?...Which of the following is not correctly matched ?...Which of the following pairs represents linkage isomers ?...The correct option(s) regarding the complex [Co(en) (NH3)3(H2O)]3+ :- (en = H2NCH2CH2NH2) is /are...Which of the following complexes are low spin and diamagnetic ?(1) K4[Os(CN)6] (2) [Mo(CO)6] (3) [Mn(CN)6]4-Select the c...