Coordination CompoundHard
Question
Which of the following is not correctly matched ?
Options
A.[Fe(en)3]3+ - sp3d2 and paramagnetic
B.[FeCl4]2- - sp3 and paramagnetic
C.[Fe(CN)6]3- - d2sp3 and paramagnetic
D.[Fe(CO)2(NO+)2] - sp3 and diamagnetic
Solution
(A) [Fe(en)3]3+ - d2sp3 with one unpaired electron. ′en′ is a strong field chelating ligand and thus compels for the pairing of electrons to have d2sp3 hybridisation. It is paramagnetic with one unpaired electron (valence shell electron configuration, 3d5)
(B) [FeCl4]2- - sp3 with four unpaired electrons. It is paramagnetic with four unpaired electrons (valence shell electron configuration, 3d6).
(C) [Fe(CN)6]3- - d2sp3 with one unpaired electron. ′CN-′ is a strong field ligand and, therefore, it compels for pairing of electrons to have d2sp3 hybridisation. It is paramagnetic with one unpaired electron (valence shell electron configuration, 3d5)
(D) [Fe(CO)2(NO+)2] - sp3 and diamagnetic. Iron is in -II oxidation state. So,
[Fe(CO)2(NO+)2]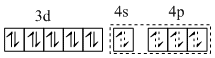
sp3hybridisation
(B) [FeCl4]2- - sp3 with four unpaired electrons. It is paramagnetic with four unpaired electrons (valence shell electron configuration, 3d6).
(C) [Fe(CN)6]3- - d2sp3 with one unpaired electron. ′CN-′ is a strong field ligand and, therefore, it compels for pairing of electrons to have d2sp3 hybridisation. It is paramagnetic with one unpaired electron (valence shell electron configuration, 3d5)
(D) [Fe(CO)2(NO+)2] - sp3 and diamagnetic. Iron is in -II oxidation state. So,
[Fe(CO)2(NO+)2]
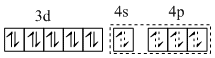
sp3hybridisation
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which is true for [Ni(en)2]2+ ? (Atomic number of nickel is 28)...The complex ion has two optical isomers. Their correct configurations are :...Which has maximum tendency to form complexes :...The EAN of platinum in potassium hexachloroplatinate (IV) is:...Which of the following is/are inner orbital complexes as well as paramagnetic with magnetic moment equal to 3.78 (approx...