Coordination CompoundHard
Question
Which of the following complexes are low spin and diamagnetic ?
(1) K4[Os(CN)6] (2) [Mo(CO)6] (3) [Mn(CN)6]4-
Select the correct answer using the codes given below.
(1) K4[Os(CN)6] (2) [Mo(CO)6] (3) [Mn(CN)6]4-
Select the correct answer using the codes given below.
Options
A.1, 2 and 3
B.1 and 2 only
C.1 and 3 only
D.2 and 3 only
Solution
All are low spin complexes but (3) has one unpaired electron so it is diamagnetic.
Mn2+, [Ar] 3d5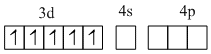
[Mn(CN)6]4-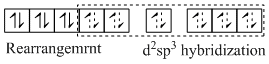
Mn2+, [Ar] 3d5

[Mn(CN)6]4-

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following pairs of name and formula of complexes, is correct ?...Lanthanide for which +II and +III oxidation states are common is :...The complex [Fe(H2O)5NO]2+ is formed in the brown ring test for nitrates when freshly prepared FeSO4 solution is added t...In Fe2(CO)9, the two iron atoms are...π-bonding is not involved in:...