Question
The correct option(s) regarding the complex [Co(en) (NH3)3(H2O)]3+ :-
(en = H2NCH2CH2NH2) is /are
Options
It has two geometrical isomers
It will have three geometrical isomers if bidentate 'en' is replaced by two cyanide ligands
It is paramagnetic
It absorbs light at longer wavelength as compared to [Co(en) (NH3)4]3+
Solution
(A) [Co(en)(NH3)3(H2O)]+3 complex is type of [M(AA)b3c] have two G.I.
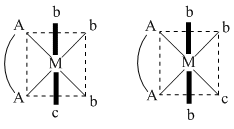
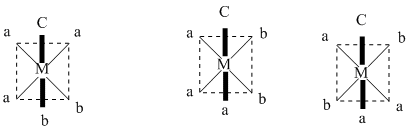
(B) If (en) is replaced by two cynide ligand, complex will be type of [Ma3b2c] and have 3 G.I
(C) [Co(en)(NH3)3(H2O)]3+ have d6 configuration () on central metal with SFL therefore it is dimagnetic in nature.
(D) Complex [Co(en)(NH3)3(H2O)]3+ have lesser CFSE () value than [Co(en)(NH3)4]3+ therefore complex [Co(en)(NH3)3(H2O)]+ absorbs longer wavelength for d–d transition.
Â
Create a free account to view solution
View Solution Free