Coordination CompoundHard
Question
The complex K4[Zn(CN)4(O2)2] is oxidised into K2[Zn(CN)4(O2)2], then which of the following is correct ?
Options
A.Zn (II) is oxidised into Zn (IV)
B.Paramagnetic moment decreases
C.O - O bond length increases
D.Paramagnetic moment increases
Solution
K4[ZnII(CN)4(O2)2]4- 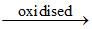 K2[ZnII(CN)4(O2)2]2-.
K2[ZnII(CN)4(O2)2]2-.
O2- (has 1unpaired electron in antibonding MO)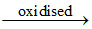 O2 (has 2 unpaired electrons in antibonding MO).
O2 (has 2 unpaired electrons in antibonding MO).
So, paramagnetic moment increases.
O2- (has 1unpaired electron in antibonding MO)
So, paramagnetic moment increases.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The number of chloride ions which would be precipitated when the complex PtCl4.4NH3 is treated with silver nitrate is:(h...A complex of platinum, ammonia and chloride produces four ions per molecule in the solution. The structure consistent wi...In which of the following pairs both the complexes do not show optical isomerism ?...Which of the following is/are correctly matched ?...The octahedral complex of a metal ion M+3 with four monodentate ligands L1, L2, L3 and L4 absorb wavelengths in the regi...