Coordination CompoundHard
Question
Which of the following is/are correctly matched ?
Options
A.[Co(PMe3)4] - sp3 and one unpaired electron.
B.[Ni(en)3](NO2)2 - sp3d2 and two unpaired electrons.
C.[V (NH3)6]Cl3- sp3d2 and two unpaired electrons.
D.[Mn (NO+)3(CO)] - sp3 and diamagnetic.
Solution
(A) [Co(PMe3)4] 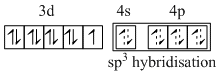
(B) [Ni(en)3]2+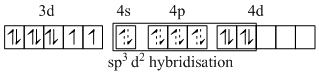
(C) [V (NH3)6]3+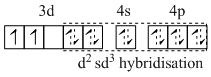
(D) [Mn3- (NO+)3 (CO)]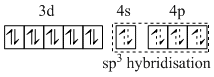
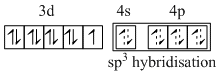
(B) [Ni(en)3]2+

(C) [V (NH3)6]3+

(D) [Mn3- (NO+)3 (CO)]
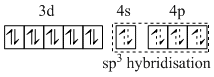
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The ligand called π acid is :...The correct structure of ethylenediaminetetraacetic acid (EDTA) is...(CFSE = Crystal Field stasilisation energy )The correct relation of x1 and x2 are :-...Which of the following statements is correct for complex [Cr(NH3)(CN)4(NO)]2 - (given that n = 1)?...Which of the following does not have optical isomer ?...