JEE Main | 2014Coordination CompoundHard
Question
The octahedral complex of a metal ion M+3 with four monodentate ligands L1, L2, L3 and L4 absorb wavelengths in the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is
Options
A.L4 < L3 < L2 < L1
B.L1 < L3 < L2 < L4
C.L3 < L2 < L4 < L1
D.L1 < L2 < L4 < L3
Solution
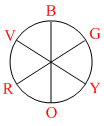
The energy of red light is less than that of violet light.
So energy order is
Red < Yellow < Green < Blue
The complex absorbs lower energy light lower will be its strength. So order of ligand strength is
L1 < L3 < L2 < L4
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is π complex:...The number of donor sites in dimethyl glyoxime, glycinato, diethylene triamine and EDTA are respectively:...The oxidation sate of iron in Na4[Fe(CN)5(NOS)] is...On adding AgNO3 solution to a solution of [Pt(NH3)3Cl3]Cl, the percentage of total chloride ion precipitated is:...Complex ion [ FeN3(O2)(SCN)4]4 - is named as : (coordination number of central metal ion in complex is six)...