Coordination CompoundHard
Question
A complex of platinum, ammonia and chloride produces four ions per molecule in the solution. The structure consistent with the observation is:
Options
A.[Pt(NH3)4]Cl
B.[Pt(NH3)2Cl4]
C.[Pt(NH3)5Cl]Cl3
D.[Pt(NH3)4Cl2]Cl2
Solution
[Pt(NH3)5Cl]Cl3 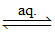 [Pt(NH3)5Cl]3+ + 3Cl-
[Pt(NH3)5Cl]3+ + 3Cl-
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is greatest paramagnetic ?...In which of the following pairs of complexes the central metals/ions do have same effective atomic number ?...For the correct assignment of electronic configuration of a complex, the valence bond theory often requires the measurem...Which of the following complexes is not a chelate ?...An aqueous solution of CoCl2 on addition of exess of concentrated HCl turns blue due to formation of :...