Coordination CompoundHard
Question
Which is true for [Ni(en)2]2+ ? (Atomic number of nickel is 28)
Options
A.Paramagnetism, dsp2, square planar, coordination number of Ni = 2
B.Diamagnetism, dsp2, square planar, coordination number of Ni = 4
C.Diamagnetism, sp3, tetrahedral, coordination number of Ni = 4
D.Paramagnetism, sp3, tetrahedral, coordination number of Ni = 4
Solution
The oxidation state of nickel is in complex = x + 4 (0) = +2 or x = +2.
Ethylenediamine is a strong field chelating ligand and so,
[Ni(en)2]2+
So, geometry is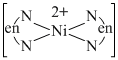
Ethylenediamine is a strong field chelating ligand and so,
[Ni(en)2]2+

So, geometry is

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which one of the following complexes in an outer orbital complex ?...Consider the following reaction and statements : [Co(NH3)4Br2]+ + Br- → [Co(NH3)3Br3] + NH3 (I) Two isomers are produ...The compound Na2IrCl6 reacts with triphenylphosphine in diethyleneglycol in an atmosphere of CO to give [IrCl(CO)(PPh3)2...Which of the following statements is not true?...Which of the following is non-conducting ?...