Coordination CompoundHard
Question
The complex ion has two optical isomers. Their correct configurations are :
Options
A.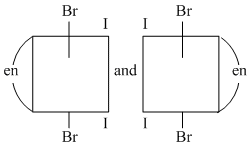

B.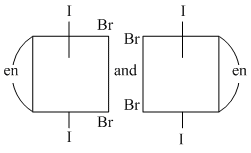

C.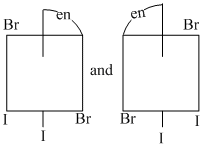

D.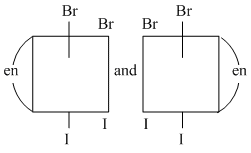

Solution
(A) Both are identical structures and each have plane of symmetry ; so they are not optical isomers.
(B) Both are identical structures and each have plane of symmetry ; so they are not optical isomers.
(C) Both are identical structures and each have plane of symmetry ; so they are not optical isomers.
(D) Both are mirror images of each other which are non-superimposable (they lack plane of symmetry)
(B) Both are identical structures and each have plane of symmetry ; so they are not optical isomers.
(C) Both are identical structures and each have plane of symmetry ; so they are not optical isomers.
(D) Both are mirror images of each other which are non-superimposable (they lack plane of symmetry)
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The complex [Fe(H2O)5NO]2+ is formed in the brown ring test for nitrates when freshly prepared FeSO4 solution is added t...The structure of iron pentacarbonyl is:...All ligands are :...Concentrated H2SO4 will not dehydrate the following complex :...NiCl2{P(C2H5)2(C6H5)}2 exhibits temperature dependent magnetic behaviour (paramagnetic/diamagnetic).The coordination geo...