Coordination CompoundHard
Question
The crystal field splitting energy for octahedral complex (ᐃo) and that for tetrahedral complex (ᐃt ) are related as:
Options
A.ᐃt = 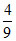 ᐃ0
ᐃ0
B.ᐃt = 0.56 ᐃ0
C.ᐃt = 0.33 ᐃ0
D.ᐃt = 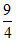 ᐃ0
ᐃ0
Solution
This may attributes to the following two reasons.
(i) There are only four ligands instead of six, so the ligand field is only two thirds the size ; as the ligand field splitting is also the two thirds the size and
(ii) the direction of the orbitals does not concide with the direction of the ligands. This reduces the crystal field splitting by roughly further two third. So ᐃt =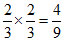 ᐃo
ᐃo
(i) There are only four ligands instead of six, so the ligand field is only two thirds the size ; as the ligand field splitting is also the two thirds the size and
(ii) the direction of the orbitals does not concide with the direction of the ligands. This reduces the crystal field splitting by roughly further two third. So ᐃt =
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following is/are correctly matched ?...The total crystal field stabilisation energy (CFSE) for the complexes [Co(H2O)6]3+ and [CoF6]3- are respectively : (P is...Complex hydrolysis of starch gives :...Complex ion [ FeN3(O2)(SCN)4]4 - is named as : (coordination number of central metal ion in complex is six)...A co-ordination complex has the formula PtCl4.2KCl. Electrical conductance measurements indicate the presence of three i...