Coordination CompoundHard
Question
A co-ordination complex has the formula PtCl4.2KCl. Electrical conductance measurements indicate the presence of three ion in one formula unit. Treatment with AgNO3 produces no precipitate of AgCl. What is the co-ordination number of Pt in this complex ?
Options
A.5
B.6
C.4
D.3
Solution
PtCl4.2KCl ≡ K2[PtCl6] 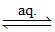 2K+ + [PtCl6]2-.
2K+ + [PtCl6]2-.
So, it has three ions per formula unit. The Cl- is present in coordination sphere so it will not give white precipitate with silver nitrate solution. So in the compound the coordination number of platinum is 6.
So, it has three ions per formula unit. The Cl- is present in coordination sphere so it will not give white precipitate with silver nitrate solution. So in the compound the coordination number of platinum is 6.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
EAN of a metal carbonyl M(CO)x is 36. If atomic number of metal M is 26, what is the value of x ?...Which of the following complex is not correctly matched with its geometry ?...Formula of ferrocene is:...Which of the following complexes has a geometry different from others ?...The number of unpaired electrons present in complex ion [FeF6]3- is :...