Coordination CompoundHard
Question
Which of the following will exhibit maximum ionic conductivity?
Options
A.K4 [Fe(CN6]
B.[Co(NH3)6] Cl3
C.[Cu(NH3)4] Cl2
D.[Ni (CO)4]
Solution
Conductivity α number of ions in the solution.
(A) K4[Fe(CN)6]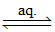 4K+ (aq) + [Fe(CN)6]4- (aq)
4K+ (aq) + [Fe(CN)6]4- (aq)
4 : 1 electrolyte.
It contains maximum number of ions i.e. 5.
(B) [Co(NH3)6] Cl3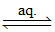 [Co(NH3)6]3+ (aq) + 3Cl- (aq)
[Co(NH3)6]3+ (aq) + 3Cl- (aq)
1 : 3 electrolyte.
(C) [Cu(NH3)4] Cl2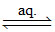 [Cu(NH3)4]2+ (aq) + 2Cl- (aq)
[Cu(NH3)4]2+ (aq) + 2Cl- (aq)
1 : 2 electrolyte.
(D) [Ni (CO)4]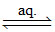 [Ni (CO)4] (aq) (neutral).
[Ni (CO)4] (aq) (neutral).
(A) K4[Fe(CN)6]
4 : 1 electrolyte.
It contains maximum number of ions i.e. 5.
(B) [Co(NH3)6] Cl3
1 : 3 electrolyte.
(C) [Cu(NH3)4] Cl2
1 : 2 electrolyte.
(D) [Ni (CO)4]
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Nickel (Z = 28) combines with a uninegative monodentate ligand X- to form a paramagnetic complex [NiX4]2-. The number of...Concentrated H2SO4 will not dehydrate the following complex :...The complex [ Fe (H2O)5 NO ]2+ is formed in the ′brown ring test′ for nitrates. Choose the incorrect stateme...The complex [Fe(H2O)5NO]2+ is formed in the brown ring test for nitrates when freshly prepared FeSO4 solution is added t...In Fe(CO)5, the Fe - C bond possesses...