Coordination CompoundHard
Question
The complex [Fe(H2O)5NO]2+ is formed in the brown ring test for nitrates when freshly prepared FeSO4 solution is added to aqueous solution of NO3- ions followed by addition of conc. H2SO4. Select correct statement about this complex.
Options
A.Hybridisation of iron is sp3d2.
B.Iron has +1 oxidation state.
C.It has magnetic moment of 3.87 b. M. confirming three unpaired electrons in Fe.
D.All the above are correct statements.
Solution
[Fe(I) (H2O)5 NO+] 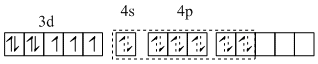
Three unpaired electrons
six - sp3d2 hybrid orbital
So m = 3.87.

Three unpaired electrons
six - sp3d2 hybrid orbital
So m = 3.87.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which is / are not correctly matched. Complex compounds IUPAC name...[Fe(en)2(H2O)2]2+ + en → complex(X). The correct statement about the complex (X) is :...Which of the following statements is correct?...Which of the following are π-bonded organometallic compounds ?...Identify the CORRECT set of details from the following :A. $\left\lbrack Co\left( {NH}_{3} \right)_{6} \right)^{3 +}$ : ...