Coordination CompoundHard
Question
Nickel (Z = 28) combines with a uninegative monodentate ligand X- to form a paramagnetic complex [NiX4]2-. The number of unpaired electron(s) in the nickel and geometry of this complex ion are, respectively
Options
A.one, tetrahedral
B.two, tetrahedral
C.one, square planar
D.two, square planar
Solution
28Ni: ......3s2, 3p6, 3d8, 4s2
Ni2+: 3s2, 3p6, 3d8
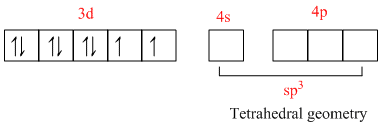
Ni2+: 3s2, 3p6, 3d8

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following complex is not correctly matched with its geometry ?...Which one is not an organometallic compound ?...Low spin complex of d6 - cation in an octahedral field will have the following energy:(ᐃ0 = Crystal Field Splittin...The EAN of metal atoms in Fe(CO)2(NO)2 and Co2(CO)8 respectively are...Which of the following species is not expected to be a ligand...