Coordination CompoundHard
Question
The complex [ Fe (H2O)5 NO ]2+ is formed in the ′brown ring test′ for nitrates. Choose the incorrect statement for the complex.
Options
A.Its magnetic moment is approximately 3.9 B.M.
B.The oxidation state of iron is + 1
C.The hybridisation of central metal ion is sp3 d2
D.The brown colour of the ring is due to d - d transition.
Solution
The iron is in +1 oxidation state with 3d7 electron configuration and NO is NO+.
[Fe (H2O)5 NO ]2+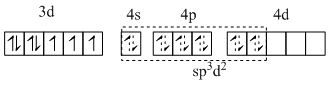
So, μ =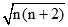 or μ =
or μ = 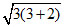 = 3.9
= 3.9
The colour is due to charge transfer not because of d-d transition of electron.
[Fe (H2O)5 NO ]2+

So, μ =
The colour is due to charge transfer not because of d-d transition of electron.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The oxidation sate of iron in Na4[Fe(CN)5(NOS)] is...The number of possible isomers of an octahedral complex [Co(C2O4)2(NH3)2]- is...Which of the following statements are correct for the complex [Co(NH3)4(Cl) (NO2)]Cl ?1. Cobalt is in +III oxidation sta...Complex ion [ FeN3(O2)(SCN)4]4 - is named as : (coordination number of central metal ion in complex is six)...The geometry of [NiCI4]2- and [NiCI2 (PMe3)2] are :...