Coordination CompoundHard
Question
Complexes [Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4 can be distinguished by :
Options
A.conductance measurement
B.using BaCl2
C.using AgNO3
D.all of these
Solution
(A) [Co(NH3)5SO4]Br 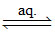 [Co(NH3)5SO4]+ (aq)+ Br- (aq)
[Co(NH3)5SO4]+ (aq)+ Br- (aq)
[Co(NH3)5Br]SO4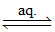 [Co(NH3)5Br]2+ (aq) + SO42- (aq)
[Co(NH3)5Br]2+ (aq) + SO42- (aq)
Both complexes have same number of ions in solutions but have different number of electrical charges. Hence they can be distinguished by conductance measurement.
(B) Br- (aq)+ Ag+ (aq) → AgBr↓ (pale yellow)
SO42- (aq.) + Ag+ → Ag2SO4 ↓ (white)
(C) Ba2+ (aq) + SO42- (aq) → BaSO4↓ (white)
Ba2+ (aq) + 2 Br- (aq) → BaBr2 (aq).
[Co(NH3)5Br]SO4
Both complexes have same number of ions in solutions but have different number of electrical charges. Hence they can be distinguished by conductance measurement.
(B) Br- (aq)+ Ag+ (aq) → AgBr↓ (pale yellow)
SO42- (aq.) + Ag+ → Ag2SO4 ↓ (white)
(C) Ba2+ (aq) + SO42- (aq) → BaSO4↓ (white)
Ba2+ (aq) + 2 Br- (aq) → BaBr2 (aq).
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The purple colour of KMnO4 is due to the transition...Which of the following statements is false?...Which of the following compounds shows optical isomerism?...Which of the following is greatest paramagnetic ?...Coordination compounds have great importance in biological systems. In this context which of the following statements is...