Coordination CompoundHard
Question
Which of the following complexes show geometrical as well as optical isomerism ?
(1) [Cr(OX)3]3-
(2) [Rh(en)2Cl2]+
(3) [Co(NH3)2(Cl)2(en)]+
Select the correct answer using the codes given below.
(1) [Cr(OX)3]3-
(2) [Rh(en)2Cl2]+
(3) [Co(NH3)2(Cl)2(en)]+
Select the correct answer using the codes given below.
Options
A.1 only
B.1 and 2 only
C.2 and 3 only
D.All 1, 2, 3
Solution
(A) With symmetrical bidentate ligands. [(Cr(ox)3]3- does no show geometrical isomerism but it can show only optical isomerism.
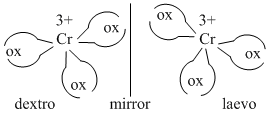
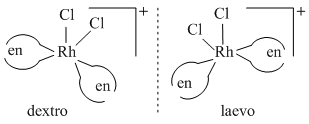
(B)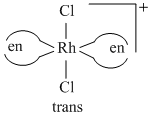
(C)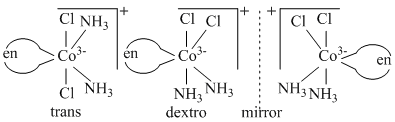

(B)


(C)

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The ligand called π acid is :...Number of bridging ligand and number of bromine atoms in compound decaammine-μ-hydroxo dichromium (III) bromide are...Which of the following is not an ore of iron ?...Among the following complexes the one which shows Zero crystal field stabilization energy (CFSE) is...Which of the following statements is/are correct ?...