Analytical ChemistryHard
Question
The solution of sodium meta aluminate on diluting with water and then boiling with ammonium chloride gives:
Options
A.[Al(H2O)5OH]2+
B.AlCl3
C.Al (OH)3
D.NaAl(OH)4
Solution
[Al(OH)4]- + NH4+ 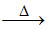 Al(OH)3 ↓ + NH3↑ + H2O
Al(OH)3 ↓ + NH3↑ + H2O
Hydroxide ion concentration is reduced owing to the formation of ammonia (a weak base) which escapes on boiling.
Hydroxide ion concentration is reduced owing to the formation of ammonia (a weak base) which escapes on boiling.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
What final product(s) is/are formed in the following series of reactions ?Concentrated borax solution + silver nitrate s...Which of the following solutions give white precipitate with Pb(NO3)2 as well as with Ba(NO3)2 ?...When H2S is passed through Hg2S we get...A metal salt solution forms a yellow precipitate with potassium chromate in acetic acid, a white precipitate with dilute...CrO42- + H+ + H2O2 X + H2OIdentify the correct statement with respect to X....