Analytical ChemistryHard
Question
CrO42- + H+ + H2O2 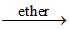 X + H2O
X + H2O
Identify the correct statement with respect to X.
Identify the correct statement with respect to X.
Options
A.It is an acid anhydride of chromic acid.
B.It is a red colour compound which can be extracted easily into the etherial phase.
C.It is chromium peroxide which produces blue colouration in etherial layer on gentle shaking.
D.It is Cr2O3 which is used as a green pigment.
Solution
CrO42- + 2H+ + 2H2O2 → CrO5 (chromium peroxide) + 3H2O
It in etherial layer develops blue colouration.
It in etherial layer develops blue colouration.
Create a free account to view solution
View Solution Free