Analytical ChemistryHard
Question
What final product(s) is/are formed in the following series of reactions ?
Concentrated borax solution + silver nitrate solution → Precipitate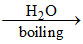 Products (final)
Products (final)
Concentrated borax solution + silver nitrate solution → Precipitate
Options
A.Ag3BO3
B.Ag2O
C.H3BO3
D.AgBO2
Solution
B4O72- + 4Ag+ + H2O → 4AgBO2 ↓ (white) + 2H+
2AgBO2 ↓ + 3H2O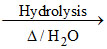 Ag2O↓ (brown) + 2H3BO3
Ag2O↓ (brown) + 2H3BO3
2AgBO2 ↓ + 3H2O
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
In II B group of basic radicals, the yellow precipitate (s) is/are given by :...A solution containing SCN- ions can be used to test one or more out of : Fe3+, Co2+ , Cu2+, Ag+ and Hg2+....Which of the following statements is/are true ?...If one strand of DNA has the sequence ATCGTATG, the sequence in the complementary strand would be :...In which of the following reactions, white precipitate is obtained as one of the reaction products ?...