Ionic EquilibriumHard
Question
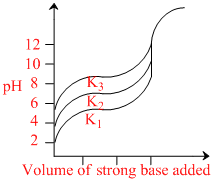
Titration curves for 0.1M solutions of three weak acids HA1, HA2 and HA3 with ionization constants K1, K2 and K3 respectively are plotted as shown in the figure. Which of the following is/are true?

Options
A.K2 = (K1 + K3)/2
B.K1 < K3
C.K1 > K2
D.K2 > K3
Solution
Higher the Ka of acid lower is the pH of acid for same concentration.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A well is dug in a bed of rock containing fluorspar (CaF2). If the well contains 20000 L of water, what is the amount of...If the solubility of lithium sodium hexafluorido aluminate, Li3Na3 (AlF6)2 is ′s′ mol lt-1, its solubility p...Which solution will have pH closer to 1.0:...An aqueous solution is prepared by dissolving 0.1 mole H2CO3 in sufficient water to get 100 ml solution at 25o C. For H2...Equimolar solutions of the following substances were prepared separately. Which one of these will record the highest pH ...