Ionic EquilibriumHard
Question
0.2 millimoles of Zn2+ ion is mixed with (NH4)2 S of molarity 0.02 M. The amount of Zn2+ remains unprecipitated in 20 mL of this solution would be (Given : KSP ZnS = 4 × 10-24)
Options
A.5.2 × 10-22 g
B.2.6 × 10-22 g
C.2 × 10-23 g
D.none of these
Solution
S2- + Zn2+ → ZnS (s)
m.moles 0.4 0.2
0.2 -
so in solution,
[S2-] =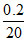 = 0.01 M
= 0.01 M
∴ [Zn2+] =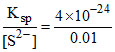 = 4 × 10-22 M
= 4 × 10-22 M
∴ mass of Zn2+ remain unprecipitated in 20 ml of solution =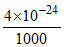 × 20 × 65 = 5.2 × 10-22 gm.
× 20 × 65 = 5.2 × 10-22 gm.
m.moles 0.4 0.2
0.2 -
so in solution,
[S2-] =
∴ [Zn2+] =
∴ mass of Zn2+ remain unprecipitated in 20 ml of solution =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?...Protective sols are :-...HCHO reacts with CH3MgI followed by hydrolysis to give :-...Which of the following is/are correct regarding buffer solution ?...When 0.1 mole solid NaOH is added in 1lt of 0.1M NH3(aq) then which statement is going to wrong?(Kb = 2 × 10-5, log...