Ionic EquilibriumHard
Question
A solution prepared by dissolving 2.8 g of lime (CaO) in enough water to make 1.00 L of lime water (Ca(OH)2(aq.)). If solubility of Ca(OH)2 in water is 1.48 gm/lt. The pH of the solution obtained will be:
[log 2 = 0.3, Ca = 40 , O = 16, H = 1]
[log 2 = 0.3, Ca = 40 , O = 16, H = 1]
Options
A.12.3
B.12.6
C.1.3
D.13
Solution
Ca(OH)2 ⇋ Ca2+ + 2OH-
t = 0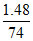 = 0.02 0 0
= 0.02 0 0
teq 0 0.02 0.04
so, pOH = 2 - log 4 = 1.4
so pH = 12.6
t = 0
teq 0 0.02 0.04
so, pOH = 2 - log 4 = 1.4
so pH = 12.6
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A volume of 250 ml of saturated clear solution of CaC2O4(aq) requires 6.0 ml of 0.001 M-KMnO4 in acidic medium for compl...Which of the following are true for an acid-base titration ?...The acid ionization constant of Zn2+ is 2.0 × 10–10. What is the pH of 0.001 M solution of ZnCl2? (log 2 = 0.3)...Degree of hydrolysis for a salt of strong acid and weak base is...20 cm3 of x M solution of HCl is exactly neutralised by 40 cm3 of 0.05 M NaOH solution, the pH of HCl solution is...