Ionic EquilibriumHard
Question
A certain mixture of HCl and CH3- COOH is 0.1 M in each of the acids. 20 ml of this solution is titrated against 0.1M NaOH. By how many units does the pH change from the start to the stage when the HCl is almost completely neutralised and acidic acid remains unreacted ? Ka for acetic acid = 2 × 10-5.
Options
A.1.5
B.3
C.2
D.3.25
Solution
m. moles of HCl = 0.1 × 20 = 2
m. moles of CH3COOH = 0.1 × 20 = 2
After titration of HCl by NaOH
[CH3COOH] =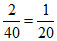 M
M
∴ pH =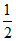 (pKa - log C) =
(pKa - log C) = 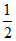 [5 - log 2 - log (
[5 - log 2 - log (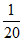 )] = 3.
)] = 3.
m. moles of CH3COOH = 0.1 × 20 = 2
After titration of HCl by NaOH
[CH3COOH] =
∴ pH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of a mixture containing 0.10 M X- and 0.20 M HX is: [pKb (X-) = 4]...100 ml of 0.5M hydrazoic acid (HN3, Ka = 3.6 × 10-4) and 400ml of 0.1M cyanic acid (HOCN, Ka = 8 × 10-4) are m...A plano convex lens fits exactly into a plano concave lens. Their plane surfaces are parallel to each other. If lenses a...Which of the following salts will give highest pH in water?...A buffer solution contains 1 mole of (NH4)2SO4 and 1 mole of NH4OH (Kb = 10-5). The pH of solution will be:...