Ionic EquilibriumHard
Question
A buffer solution contains 1 mole of (NH4)2SO4 and 1 mole of NH4OH (Kb = 10-5). The pH of solution will be:
Options
A.5
B.9
C.5.3
D.8.7
Solution
pOH = pKb + log 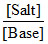 = pKb + log
= pKb + log 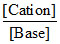
[NH4+] = 2 × mole of (NH4)2SO4 ∴ pOH = 5 + log 2 = 5.3]
or pH = 8.7
[NH4+] = 2 × mole of (NH4)2SO4 ∴ pOH = 5 + log 2 = 5.3]
or pH = 8.7
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of 0.01 M-(NH4)2SO4 and 0.02 M-NH4OH buffer (pKa of NH4+ = 9.26) is...0.1 millimole of CdSO4 are present in 10 mL acid solution of 0.08 N HCl. Now H2S is passed to precipitate all the Cd2+ i...When weak base solution (50 mL of 0.1 N NH4OH) is titrated with strong acid (0.1 N HCl), the pH of the solution initiall...Nitration of aniline in strong acidic medium also gives m-nitroaniline because...Biodegradable polymer which can be produced fromglycine and aminocaproic acid is :-...