Ionic EquilibriumHard
Question
The solubility product of AgCl is 1.8 × 10-10. Precipitation of AgCl will occur only when equal volumes of solutions of
Options
A.10-4M Ag+ and 10-4 M Cl- are mixed.
B.10-7 M Ag+ and 10-7 M Cl- are mixed.
C.10-5 M Ag+ and 10-5 M Cl- are mixed.
D.2 × 10-5 M Ag+ and 2 × 10-5 M Cl- are mixed.
Solution
Ksp = 1.8 × 10-10
Precipitation of AgCI will occuir only where
KIP (AgCI) > Ksp
KIP =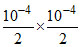 = 2.5 × 10-9 KIP > KSP precipitate is formed.
= 2.5 × 10-9 KIP > KSP precipitate is formed.
Precipitation of AgCI will occuir only where
KIP (AgCI) > Ksp
KIP =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...Select the correct order :-...0.2 millimoles of Zn2+ ion is mixed with (NH4)2 S of molarity 0.02 M. The amount of Zn2+ remains unprecipitated in 20 mL...Choose the correct statement:...Which of the following statements are true for a solution saturated with AgCI and AgBr if their solubilities in mol per ...