Ionic EquilibriumHard
Question
A buffer solution is prepared by mixing ′a′ moles of CH3COONa and ′b′ moles of CH3COOH such that (a + b) = 1, into water to make 1L buffer solution. If the buffer capacity of this buffer solution is plotted against moles of salt CH3COONa (a) then the plot obtained will be (to the scale) approximately. (As shown in figure in options)
Options
A.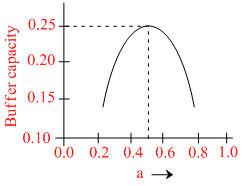

B.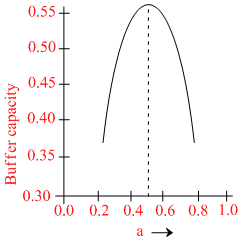

C.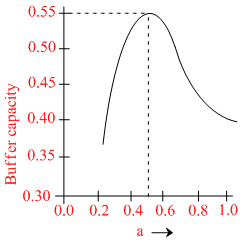

D.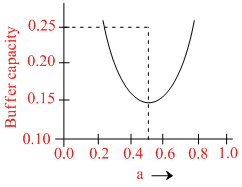

Solution
Maximum buffer capacity of a solution is given by, buffer capacity = 2.303 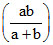 Hence the result.
Hence the result.
and a = b = 0.5, BC = 2.303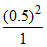 = 0.57
= 0.57
and a = b = 0.5, BC = 2.303
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At 80oC, distilled water (H3O+) has concentration equal to 1 × 10-6 mole/liter. the value of Kw, at this tempreatur...Which set is not correct for the solubility product (KSP), solubility (sg/litre) of sparingly soluble salt A3B2 (mol. wt...Which is most stable ?...Two buffer solutions, A and B, each made with acetic acid and sodium acetate differ in their pH by one unit, A has salt ...Which one of the following substances has the highest proton affinity?...