Ionic EquilibriumHard
Question
Which set is not correct for the solubility product (KSP), solubility (sg/litre) of sparingly soluble salt A3B2 (mol. wt. M) in water:
1. KSP = 108s5 2. KSP =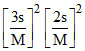 3. KSP = [1A2+]3 [2B3-]2 4. [B3-] =
3. KSP = [1A2+]3 [2B3-]2 4. [B3-] = 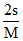 5.
5. 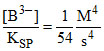
1. KSP = 108s5 2. KSP =
Options
A.1, 3
B.1,3, 5
C.2, 3, 4
D.2, 4, 5
Solution
A3B2 ⇋ 3A2+ + 2B3+
KSP = [A2+]3[B3+]2.
KSP = (3s)3 (2s)2 = 108s2
KSP = [A2+]3[B3+]2.
KSP = (3s)3 (2s)2 = 108s2
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following may act as buffer in aqueous solution?...Amongst the following hydroxides, the one which has the lowest value of Ksp at ordinary temperature (about 25oC) is:...Morphine (C17H19NO3) is administered medically to relieve pain. It is a naturally occurring base or alkaloid. What is th...In which of the following solutions, the solubility of AgCN will be greater than that in pure water :Given Ksp(AgCN) = 4...In which of the following cases, pH is greater than 7 ?...