Ionic EquilibriumHard
Question
pH of 0.01 M HS- will be:
Options
A.pH = 7 + 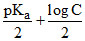
B.pH = 7 - 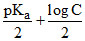
C.pH = 7 + 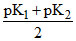
D.pH = 7 + 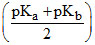
Solution
HS- + H2O ⇋ H2S + OH-
∴ [OH-] = Ch =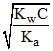 ∴ [H+] =
∴ [H+] = 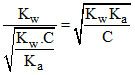
or pH = 1/2[pKw + pKa + logC]
∴ [OH-] = Ch =
or pH = 1/2[pKw + pKa + logC]
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Pure water is added into the following solutions causing a 10% increase in volume of each. The greatest % change in pH w...The pH of 0.1 M solution of the following salts increases in the order:...For ortho phosphoric acid,H3PO4 (aq) + H2O (aq) ⇋ H3O+ (aq) + H2PO4- (aq) ; Ka1H2PO4 (aq) + H2O (aq) ⇋ H3O+ ...The dissociation constant of NH3 at 27°C from the following data:NH3 + H+ $\rightleftharpoons$ NH4+; ∆H° = –52.21 kJ/mo...Which shows the maximum rate of alkaline hydrolysis :-...