Ionic EquilibriumHard
Question
The ratio of dissociation constant of two weak acids HA and HB is 4. At what moar concentration ratio, the two acids will have same pH in separate solutions:
Options
A.2
B.0.5
C.4
D.0.25
Solution
Ka = 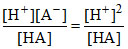 (∴ [H+] = [A-])
(∴ [H+] = [A-])
and Kb =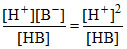 (∴ [H+] = [B-])
(∴ [H+] = [B-])
Also H+ are same ∴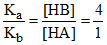
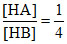
and Kb =
Also H+ are same ∴
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which one of the following statements is not true?...A volume of 20 ml of 0.8 M-HCN solution is mixed with 80 ml of 0.4 M-NaCN solution. Calculate the pH of the resulting so...For a tribasic acid, H3A, Ka1 = 2 × 10−5, Ka2 = 5 × 10−9 and Ka3 = 4 × 10−12. The value of $\frac{\left\lbrack A^{3 -} \...Which is/are correct statements : (i) In any strong acid’s solution, the concentration of [OH-] will be zero. (ii) If DG...The solubility of A2X3 is y mol dm–3. It solubility product is...