Ionic EquilibriumHard
Question
A weak acid, HA is found to be 10% ionized in 0.01 M aqueous solution. Calculate thepH of a solution which is 0.1 M in HA and 0.05 M in NaA.
Options
A.5.365
B.6.355
C.3.653
D.6.593
Solution
α = 0.1,
Ka =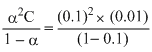 = 1.11 × 10-4
= 1.11 × 10-4
Now pKa = - log 1.11 × 10-4 = 3.9542
pH = pKa + log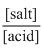
= 3.9542 + log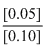 = 3.65
= 3.65
Ka =
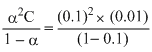 = 1.11 × 10-4
= 1.11 × 10-4Now pKa = - log 1.11 × 10-4 = 3.9542
pH = pKa + log
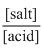
= 3.9542 + log
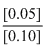 = 3.65
= 3.65Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A solution having hydrogen ion concentration is 0.0005 g eqvt./litre, its pOH is :...The indicator constant for an acidic indicator, HIn is 5 × 10−6 M. This indicator appears only in the colour of acidic f...When 20 ml of 0.2 M-DCl solution is mixed with 80 ml of 0.1 M-NaOD solution, pH of the resulting solution becomes 13.6. ...0.2 millimoles of Zn2+ ion is mixed with (NH4)2 S of molarity 0.02 M. The amount of Zn2+ remains unprecipitated in 20 mL...A buffer solution is prepared in which the concentration of NH3 is 0.30 M and the concentration of NH4+ is 0.20 M. If th...