Ionic EquilibriumHard
Question
When 20 mL of M/20 NaOH is added to 10 mL of M/10 HCl, the resulting solution will
Options
A.Turn blue litmus red.
B.Turn phenolpthalein solution pink.
C.Turns methyl orange red.
D.Will have no effect on either red or blue litmus.
Solution
mEq of NaOH = 20× 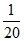 = 1
= 1
mEq of HCl = 10 ×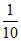 = 1
= 1
So, salt of SA/SB (NaCl) will be formed which do not hydrolyse and pH = 7. So at pH = 7, litmus has no effect. Since, blue litmus turns red a acidic solution, and red litmus turns blue, a basic solution.
i. Phenolphthalein solution turns pink a basic solution of pH > 7.
ii. Methyl orange turns red a acidic solution of pH < 7.
mEq of HCl = 10 ×
So, salt of SA/SB (NaCl) will be formed which do not hydrolyse and pH = 7. So at pH = 7, litmus has no effect. Since, blue litmus turns red a acidic solution, and red litmus turns blue, a basic solution.
i. Phenolphthalein solution turns pink a basic solution of pH > 7.
ii. Methyl orange turns red a acidic solution of pH < 7.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
1.4 g of an organic compound was digested according to Kjeldahl′s method and the ammonia evolved was absorbed in 6...The silver ion concentration in a 0.2 M solution of Ag(NH3)2NO3 is (Kdiss = 6.8 × 10−8, 1.53 = 3.4)...Which would decrease the pH of 25 ml of a 0.01 M solution of hydrochloric acid? The addition of...If 50 ml of 0.2 M KOH is added to 40 ml of 0.5 M HCOOH. the pH of the resulting solution is: (Ka = 1.8 × 10-4, log ...A 0.1 mole of AgNO3 is dissolved in 1 L of 1 M-NH3. If 0.01 mole of NaCl is added to this solution, will AgCl(s) precipi...