Ionic EquilibriumHard
Question
The dissociation constant of acetic acid at a given temperature is 1.69 × 10-5. The degree of dissociation of 0.01 M acetic acid in the presence of 0.01 M HCl is equal to
Options
A.0.41
B.0.13
C.1.69 × 10-3
D.0.013.
Solution
CH3COOH (aq) ⇋ H+ (aq) + CH3COO- (aq)
t = 0 0.01
t = eq 0.01 - x x x
[H+] = x + 0.01 ≈ 0.01 M
∴ Ka =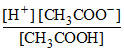 ⇒ 1.69 × 10-5 =
⇒ 1.69 × 10-5 = 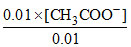
∴ [CH3COO-] = 1.69 × 10-5 M
So, degree of dissociation of CH3COOH =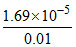 = 1.69 × 10-3
= 1.69 × 10-3
t = 0 0.01
t = eq 0.01 - x x x
[H+] = x + 0.01 ≈ 0.01 M
∴ Ka =
∴ [CH3COO-] = 1.69 × 10-5 M
So, degree of dissociation of CH3COOH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
An aqueous solution contains 0.02 M-FeCl2 and 0.05 M-FeCl3. The solubility products are 8 × 10−16 for Fe(OH)2 and 4 × 10...When weak base solution (50 ml of 0.1 N NH4OH) is titrated with strong acid 0.1 N HCl, the pH of solution initially decr...A well is dug in a bed of rock containing fluorspar (CaF2). If the well contains 20000 L of water, what is the amount of...Which of the following the weakest base is :...Which one is the correct graph (figure) for the corresponding acid base titration?...